AXILD model: Efficacy testing of new drug candidates for pulmonary fibrosiser
L.L. de Maddalena, N. Albrecher, G. Raggi, L. Froment, A. Cagnan, N. Roldan, G. Marchini, N. Hobi
Interstitial Lung Diseases (ILD) and Idiopathic Pulmonary Fibrosis (IPF)
Interstitial lung diseases are a group of disorders characterized by progressive scarring of the lung tissue. One of the most common and severe forms is Idiopathic Pulmonary Fibrosis, where the increase of thickness and stiffness of the tissue lead to impaired of lung function. Even if there are currently two drugs available to slow down the disease progression, Pirfenidone and Nintedanib, none of them allow to reverse the damage caused by IPF.
Modelling ILD using our AXBarrier-on-chip system
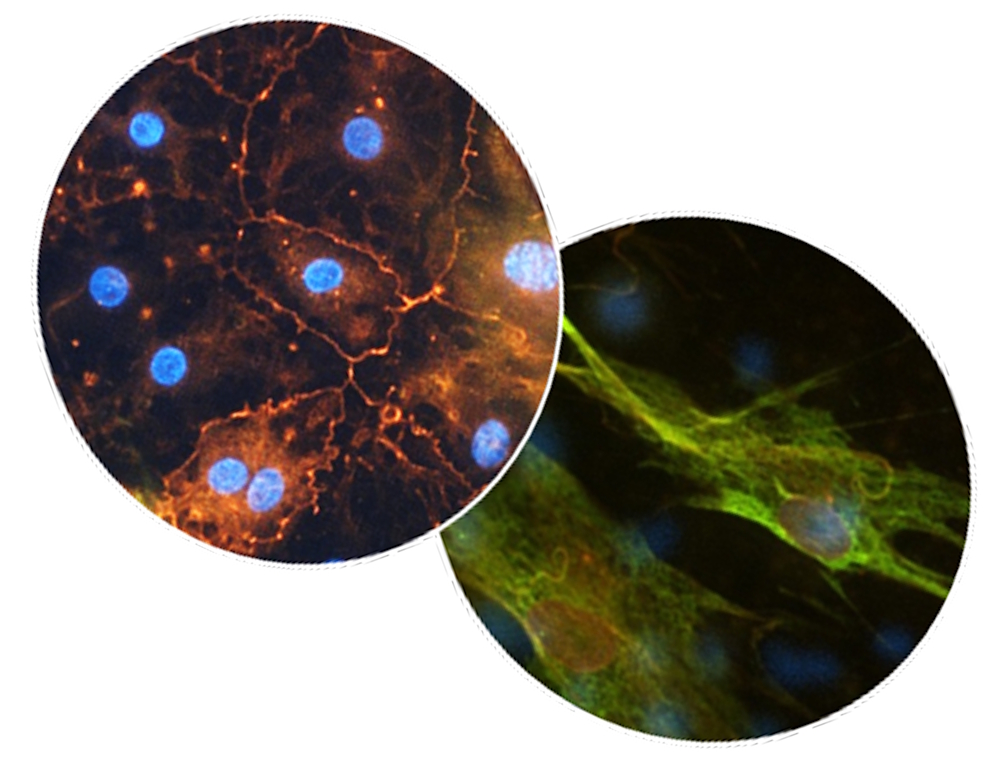
This study details the development of an in vitro ILD model using primary human alveolar epithelial cells co-cultured with healthy human lung fibroblasts on our AX12 plate. TGF-β1, a key pro-fibrotic cytokine, was introduced to trigger hallmark features of lung fibrosis. As a proof of concept for drug evaluation, Nintedanib was tested for its antifibrotic effects.
Key findings:
- TGF-ß1 stimulation led to upregulation of fibrotic markers at both protein and gene levels, increased extracellular matrix (ECM) deposition, and elevation of wound healing associated factors.
- Fibroblast activation and lung fibrosis biomarkers were upregulated upon TGF- ß1 treatment
- Nintedanib partially counteracted the effects of TGF-β, reducing fibroblast activation, ECM deposition decreased biomarker secretion.
Conclusion
Our AXILD model provides a relevant and reliable platform to study the mechanisms underlying interstitial lung diseases and to evaluate the safety and efficacy of new therapeutic candidates in a human-relevant environment.
