Patient derived AXLung-on-chip model: mimicking Interleukin-2 – induced vascular leak syndrome and patient-specific responses
Giulia Raggi, Lauriane Cabon, Laurène Froment, Nicole Albrecher, Linda Steinacher, Nina Hobi
Interleukin-2: an efficacious cancer immunotherapy with serious side effects
While Interleukin-2 (IL-2, Proleukin®) was the first approved immunotherapy for metastatic melanoma and renal cell carcinoma, its clinical use has been limited by significant side effects. Around one-third of patients treated with IL-2 develop vascular leak syndrome (VLS) — a condition that can cause pulmonary edema, inflammation, and immune cell infiltration. This challenge has driven efforts to develop safer IL-2 variants with reduced toxicity and enhanced anti-tumour efficacy.
Benchmarking IL2 variants on our patient-derived AXLung-on-chip model
To replicate IL-2-induced toxicity and patient variability, we used our AXBarrier-on-chip where patient-derived primary alveolar epithelial cells from multiple donors were in triple-culture with endothelial cells and peripheral blood mononuclear cells (PBMC) . The proinflammatory molecule TNFα was administered as positive control for inducing VLS hallmarks.
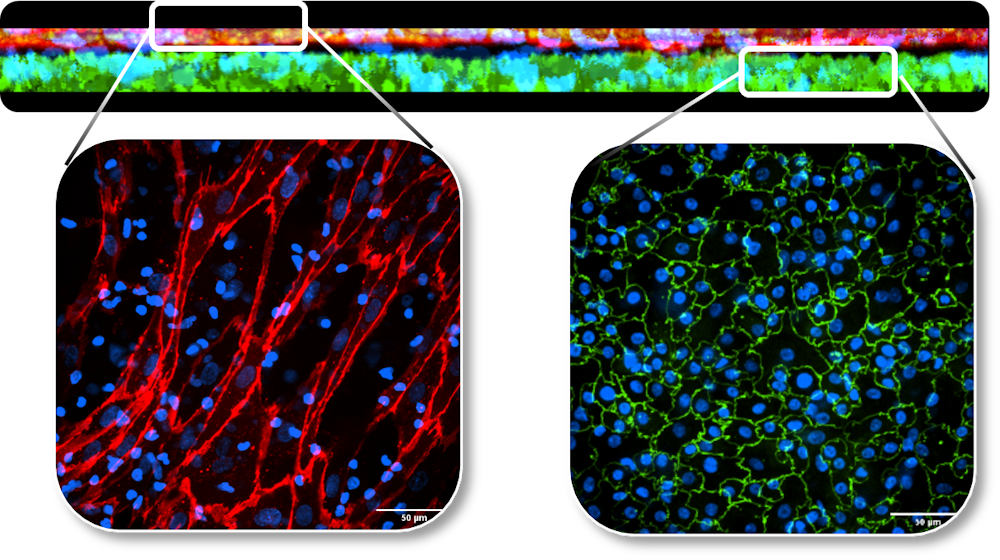
Key findings:
- IL-2 treatment at physiological doses induced VSL in a subset of donors, shown by barrier impairment and cytokine upregulation. In contrast, other donors showed no adverse effects, reflecting clinical variability observation
- Flow cytometry of PBMC harvested from the chips showed clear T-Cell and NK activation following IL-2 exposure.
- Among others, the cytokine IP-10 was significantly upregulated in response to IL-2 treatment, mirroring clinical findings and reinforcing its potential as a biomarker of IL-2–induced vascular injury
- TNFα instead, caused consistent barrier damage and inflammation across all donors, highlighting the specificity of IL-2’s variable response
Conclusion:
These results highlight the predictive potential of the AXLung-on-chip model to assess safety of immunotherapies while capturing donor-to-donor variability. It could thus contribute significantly to advancing cancer immunotherapy drug development by facilitating the selection of drugs with a more favorable benefit-risk ratio and helping to advance personalized medicine.
