Patient derived AXLung-on-chip model for immunotherapy safety: a case study with T-Cell bispecific antibodies (TCBs)
Giulia Raggi*, L. Froment, V. Micallef, T. Imler, N. Roldan, L. Cabon, N. Hobi
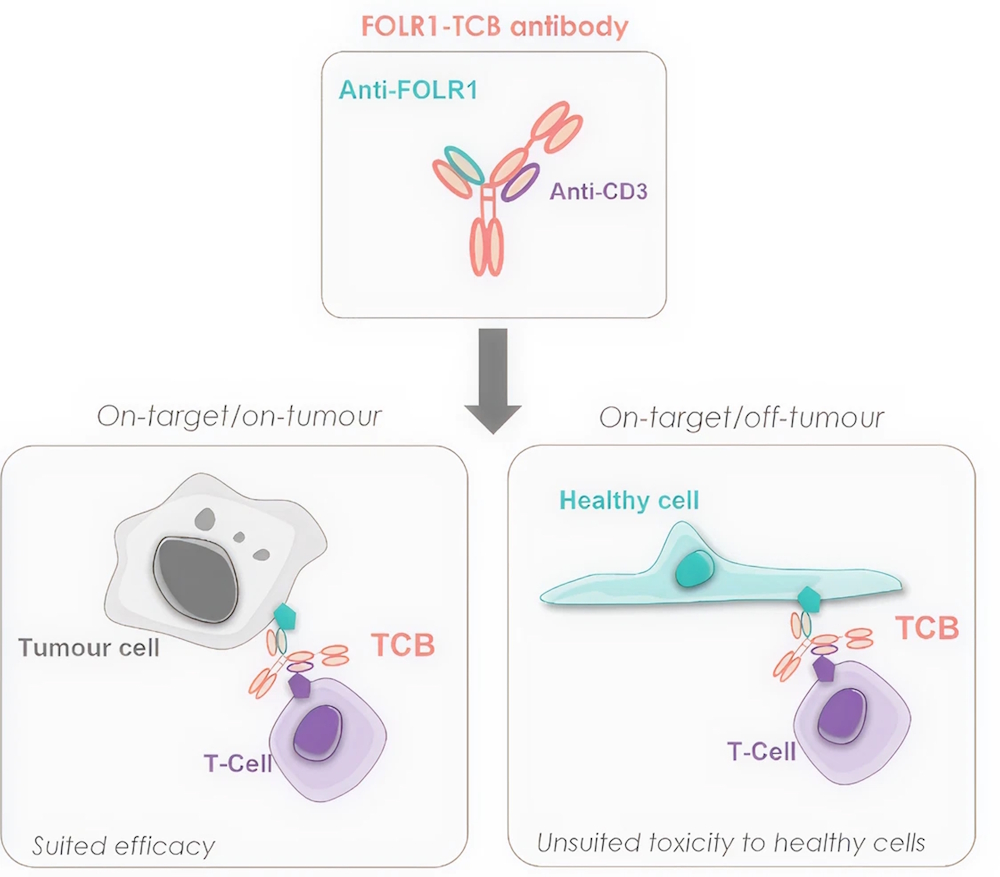
T-Cell Bispecific antibodies (TCBs): a promising immunotherapy
Immunotherapy has revolutionized cancer treatment by harnessing the body’s own immune system to recognize and destroy tumour cells. Among these approaches, T-Cell bispecific antibodies (TCBs) have emerged as a powerful new class of therapeutics. TCBs are engineered to bind both to a T-Cell and to a specific antigen on a target cell, leading to T-Cell–mediated killing of cells expressing the target. Despite their potential, such treatments present a high risk of immune related adverse events, including on-target/off-tumour toxicity when they bind to healthy tissues that express the same target.
Evaluating immunotherapy toxicity with a patient-derived AXLung-on-chip
In this study, we evaluated the lung specific toxicity of a Folate Receptor 1 (FOLR1)-TCB using our patient-derived AXLung-on-chip platform. FOLR1 is overexpressed in various types of cancer (including ovarian, lung, breast), making it a suitable immunotherapy target. However, it is also present in healthy epithelium, such as lung and kidney, raising concerns about off-tumor toxicity. To investigate this risk, we co-cultured primary alveolar epithelial cells from multiple donors with immune cells on our ultra-thin porous membrane of the AX12 chip.
Key findings:
- FOLR1 expression was confirmed and quantified at both gene and protein levels, comparing native lung tissue and our primary epithelial cells grown on-chip.
- FOLR1-TCB induced a pronounced on-target/off-tumour toxicity in the alveolar model as shown by increased cytotoxicity (LDH), barrier disruption (TER), increase of pro-inflammatory cytokine release (e.g., IL-6, Granzyme B) and enhanced apoptosis in epithelial cells.
- The treatment also triggered immune cell recruitment to the epithelium confirmed via live imaging; and T-Cell activation, validated by flow cytometry.
Conclusion:
Our patient-derived alveolar AXLung-on-chip platform reliably detects off-tumor immune-related toxicity associated with TCBs. This human-relevant model offers a powerful non-animal alternative for preclinical safety evaluation of next-generation immunotherapies.
