Jan 04, 2023
The FDA Modernization Act 2.0 passed
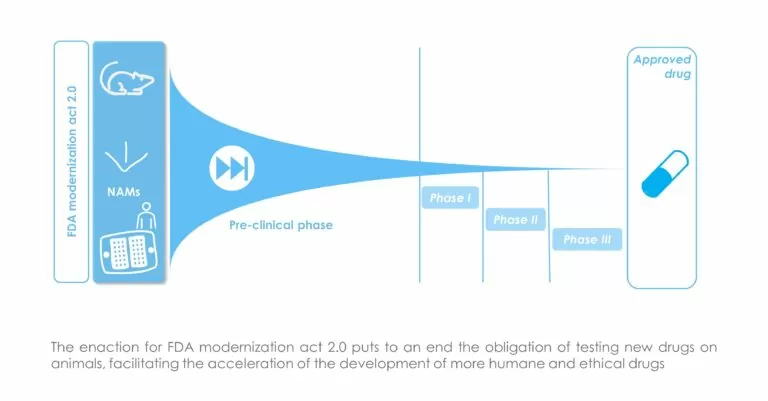
🎉 Let’s start 2023 with good news: The FDA Modernization Act 2.0 has passed congress and is inked by President Biden!
🧫 This big achievement lets the FDA consider non-animal cutting-edge testing methods as standalone tests for preclinical studies, finally allowing the use Organs-on-Chip and NAMs for drug safety and efficacy assessment.
📑 𝘛𝘩𝘦 𝘍𝘋𝘈 𝘔𝘰𝘥𝘦𝘳𝘯𝘪𝘻𝘢𝘵𝘪𝘰𝘯 𝘈𝘤𝘵 2.0 𝘱𝘳𝘰𝘮𝘪𝘴𝘦𝘴 𝘵𝘰 𝘳𝘦𝘣𝘰𝘰𝘵 𝘢 𝘣𝘳𝘰𝘬𝘦𝘯 𝘥𝘳𝘶𝘨 𝘥𝘦𝘷𝘦𝘭𝘰𝘱𝘮𝘦𝘯𝘵 𝘱𝘢𝘳𝘢𝘥𝘪𝘨𝘮 𝘢𝘯𝘥, 𝘪𝘧 𝘪𝘵 𝘪𝘴 𝘷𝘪𝘨𝘰𝘳𝘰𝘶𝘴𝘭𝘺 𝘪𝘮𝘱𝘭𝘦𝘮𝘦𝘯𝘵𝘦𝘥, 𝘸𝘪𝘭𝘭 𝘥𝘦𝘭𝘪𝘷𝘦𝘳 𝘴𝘢𝘧𝘦𝘳, 𝘮𝘰𝘳𝘦 𝘦𝘧𝘧𝘦𝘤𝘵𝘪𝘷𝘦, 𝘮𝘰𝘳𝘦 𝘳𝘦𝘭𝘪𝘢𝘣𝘭𝘦 𝘱𝘢𝘭𝘭𝘪𝘢𝘵𝘪𝘷𝘦𝘴 𝘢𝘯𝘥 𝘤𝘶𝘳𝘦𝘴 𝘵𝘰 𝘱𝘦𝘰𝘱𝘭𝘦 𝘪𝘯 𝘯𝘦𝘦𝘥. More info in the article.
Article
𝐖𝐡𝐚𝐭 𝐚𝐛𝐨𝐮𝐭 𝐲𝐨𝐮: 𝐚𝐫𝐞 𝐲𝐨𝐮 𝐫𝐞𝐚𝐝𝐲 𝐭𝐨 𝐬𝐭𝐚𝐫𝐭 𝐮𝐬𝐢𝐧𝐠 𝐎𝐫𝐠𝐚𝐧𝐬-𝐨𝐧-𝐂𝐡𝐢𝐩 ❓